Upcycling Industrial Wastewater: Recovering Phosphates for Reuse
Every year, billions of liters of industrial wastewater are discharged into rivers, lakes, and coastal waters carrying an invisible cargo that troubles environmental regulators worldwide: dissolved phosphates. Originating from fertilizer production, food processing, semiconductor manufacturing, and a dozen other industries, these compounds are simultaneously a serious ecological threat and a squandered economic opportunity. Phosphorus, the element at the heart of every phosphate molecule, is a finite, non-renewable resource with no synthetic substitute — and global reserves are quietly dwindling.
The conventional response has been treatment and disposal, including chemical precipitation, biological removal, and ultimately landfilling. But a new generation of engineers, chemists, and industrial ecologists is asking a different question. What if, instead of paying to destroy this phosphorus, we recovered it and sold it back into the supply chain?
This is the promise of industrial phosphate upcycling — and it is moving fast from laboratory curiosity to real-world deployment.
Why Phosphorus Scarcity Demands a Circular Approach
Phosphorus is one of the three macronutrients essential to all life on Earth — alongside nitrogen and potassium — and it underpins global food production. Unlike nitrogen, which can be fixed from the atmosphere, phosphorus must be mined from phosphate rock deposits concentrated in a handful of countries, most notably Morocco, China, and Russia. The European Union has listed phosphate rock as a critical raw material since 2014, reflecting growing anxiety about supply concentration and long-term availability.
According to estimates from the Global Phosphorus Research Initiative, at current extraction rates, economically viable reserves could be depleted within the next 50 to 100 years. More troubling still, the phosphorus cycle is almost entirely linear: rock is mined, converted into fertilizer, absorbed by crops, consumed by humans or livestock, and then discharged into wastewater streams. Less than 20% of the phosphorus extracted from the ground ever makes it back into agricultural soil.
At the same time, the phosphorus in wastewater is actively harmful. Even at low concentrations — as little as 0.1 milligrams per liter — phosphates can trigger eutrophication in freshwater systems, promoting explosive algal blooms that deplete oxygen, kill fish, and contaminate drinking water. Regulatory pressure from the EU's Urban Wastewater Treatment Directive and equivalent legislation in North America and Asia is making costly treatment unavoidable. The logic of recovery rather than removal is becoming impossible to ignore.
The Industrial Sources: Where the Phosphate Actually Comes From
Municipal sewage often dominates the public conversation around phosphorus pollution, but industrial effluents are equally significant — and in many respects more tractable because their flows are concentrated, controlled, and consistent. Key industrial point sources include:
Fertilizer and agrochemical manufacturing: Production washwaters and process streams from fertilizer plants carry some of the highest phosphate concentrations encountered in industrial settings — sometimes exceeding several thousand milligrams per liter.
Food and beverage processing: Dairy operations, breweries, potato processors, and meat-packing facilities generate phosphate-rich effluent from cleaning agents, naturally occurring organic phosphorus in raw materials, and additive-laden process water.
Semiconductor fabrication: Phosphoric acid is a standard etchant in chip manufacturing. Rinse waters from fabs contain dissolved orthophosphate at concentrations that are both environmentally problematic and — given the purity of the source — remarkably recoverable.
Metal finishing and surface treatment: Phosphating baths used to prepare steel and aluminum surfaces for painting produce spent liquors loaded with iron or zinc phosphate compounds that present both a hazardous waste challenge and a recovery opportunity.
Recovery Technologies: From Struvite Crystallization to Membrane Systems
The engineering toolbox for phosphate recovery has expanded significantly in the last decade. No single technology dominates; the best choice depends on influent concentration, competing ions, the desired end product, and capital constraints.
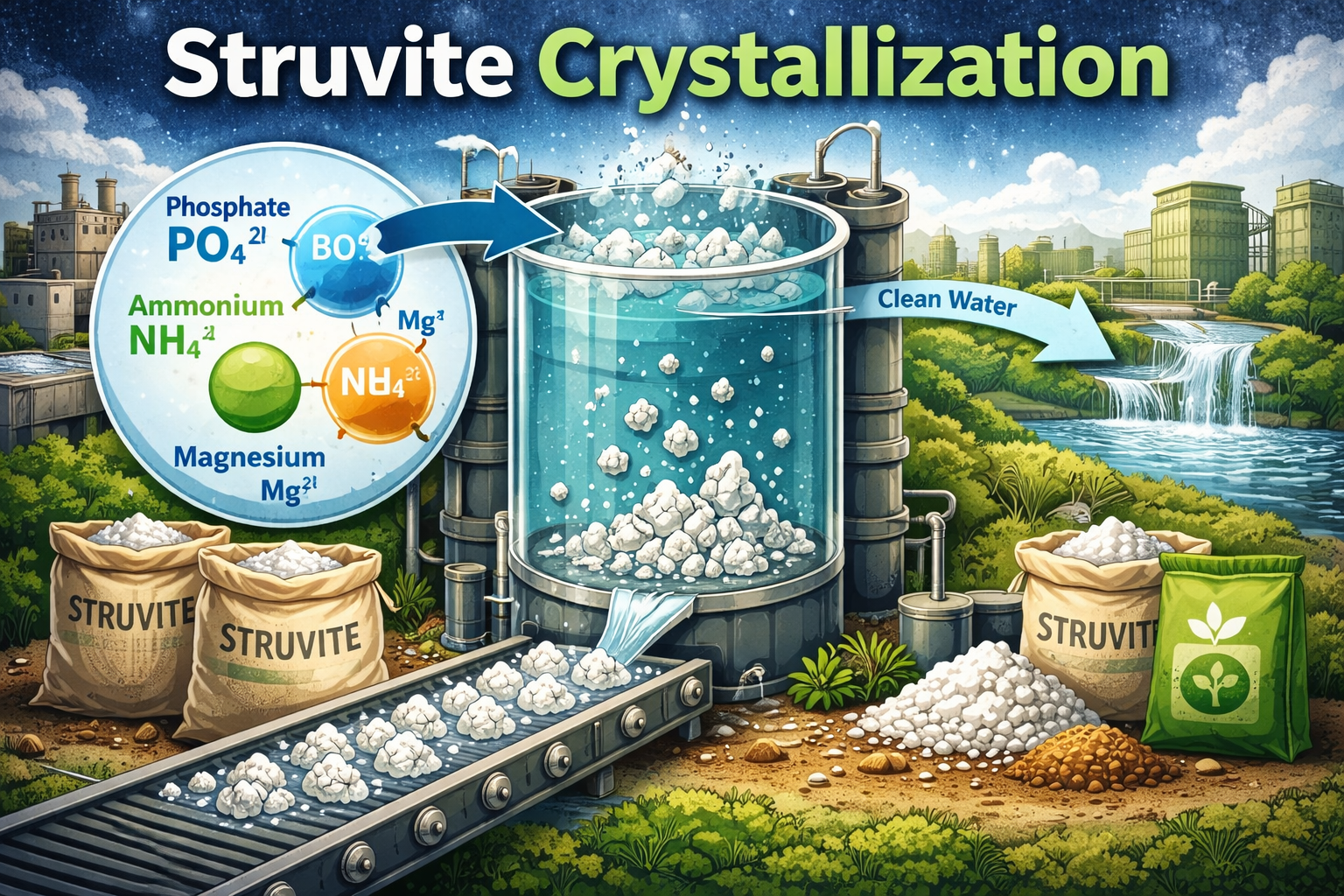
Struvite Crystallization
Struvite (magnesium ammonium phosphate, MgNH₄PO₄·6H₂O) is perhaps the most commercially mature recovery pathway. When magnesium is added to phosphate and ammonia-rich wastewater under controlled pH conditions, struvite precipitates as compact, slow-release crystals. These crystals are a recognized fertilizer product, sold commercially under brand names such as Crystal Green by Ostara. Struvite crystallization is now deployed at scale in wastewater treatment plants across North America and Europe, and industrial operators are increasingly adapting reactor designs to their specific effluent chemistries.
Calcium Phosphate Precipitation
At high pH, the addition of calcium ions (typically from lime) drives the formation of calcium phosphate phases ranging from brushite to hydroxyapatite. The resulting precipitate can, after appropriate processing, substitute for mined phosphate rock in fertilizer production. This pathway is especially relevant for streams with high phosphate concentrations and relatively low ammonia content, where struvite would be less favored thermodynamically.
Ion Exchange and Adsorption
Selective adsorbents — including engineered lanthanum-doped materials, layered double hydroxides, and biochar composites — can capture phosphate from dilute streams where precipitation is thermodynamically unfavorable. After saturation, the adsorbent is regenerated with a concentrated alkali or salt solution, producing a phosphate-rich eluate that can be further processed. This approach is gaining traction for treating semiconductor rinse waters and agricultural run-off, where concentrations are too low for direct precipitation.
Membrane Filtration and Electrodialysis
Nanofiltration and reverse osmosis membranes can concentrate phosphate-laden streams, producing a retentate suitable for crystallization alongside a clean permeate for reuse or discharge. Electrodialysis — using ion-selective membranes and an applied electric field — goes further, selectively migrating phosphate ions across the membrane into a concentrated product stream. While energy-intensive, electrodialysis is particularly attractive for high-value, high-purity applications like recovering reagent-grade phosphoric acid from semiconductor waste streams.
Closing the Loop: How Recovered Phosphate Re-enters the Supply Chain
Recovery technology is only half the story. For upcycling to be meaningful, the recovered material must displace virgin resource extraction — not simply add a layer of processing before landfill. The pathways back into productive use depend heavily on product quality.
Recovered struvite and calcium phosphates, when produced under well-controlled conditions, meet the quality criteria for use as slow-release fertilizers. Field trials in Germany, the Netherlands, and Canada have demonstrated agronomic performance comparable to mined superphosphate, with the added benefit of lower cadmium contamination — a persistent concern with some phosphate rock deposits from North Africa.
In the food and pharmaceutical sectors, high-purity recovered phosphoric acid is finding acceptance as a process chemical, closing a loop that was previously impossible without rigorous purification. Meanwhile, the European Union's revised Fertilizing Products Regulation, which came into force in 2022, explicitly creates a regulatory pathway for recovered phosphate products to be placed on the single market, removing a previously significant barrier to commercialization.
Economics: The Business Case for Phosphate Upcycling
Critics have long argued that phosphate recovery makes environmental sense but poor commercial sense. That calculus is shifting. The economics are driven by three converging forces: rising disposal and compliance costs, increasing phosphate rock prices (which spiked sharply following supply disruptions in 2021 and 2022), and the emergence of ESG-driven procurement policies that create premium markets for circularly sourced materials.
A detailed lifecycle cost analysis published in the journal Resources, Conservation and Recycling in 2023 found that struvite recovery systems at medium-to-large food processing facilities could achieve payback periods of three to seven years under current market conditions — before accounting for carbon credits or environmental compliance savings. For semiconductor fabs, where the alternative is expensive, hazardous waste disposal, the economics can be even more compelling.
Industrial symbiosis models are also emerging, in which several smaller facilities co-invest in a shared phosphate recovery hub, spreading capital costs while achieving the treatment volumes needed for economical operation. Industrial parks in the Netherlands, Denmark, and Belgium have pioneered this model, and similar initiatives are being explored in South Korea's electronics manufacturing clusters.
Challenges Still to Overcome
For all its promise, industrial phosphate upcycling is not without obstacles. Regulatory frameworks in many jurisdictions still classify recovered phosphate materials as waste rather than products, creating legal barriers to sale even when purity is demonstrably high. The EU's regulatory reforms are leading the way, but national implementations are uneven and slow.
Technical challenges persist as well. Real industrial effluents are complex mixtures containing heavy metals, organic compounds, and competing anions that can interfere with precipitation kinetics or contaminate the recovered product. Process upsets — common in batch manufacturing environments — can deliver shock loads of contaminants that destabilize recovery systems designed for steady-state operation.
Market acceptance for recovered materials also requires sustained effort. Fertilizer manufacturers and farmers accustomed to conventional superphosphate need assurance about consistency, solubility, and micronutrient content — assurances that only come with time, certification schemes, and field data. Building that trust is as important as the engineering.
The Road Ahead: Policy, Innovation, and Industry Leadership
The momentum behind phosphate upcycling is unmistakable. The European Green Deal and its Farm to Fork strategy have placed nutrient recovery at the center of agricultural and industrial policy. The United States Environmental Protection Agency is updating phosphorus numeric criteria for freshwater systems, tightening discharge limits in ways that will make recovery economically coercive for many industries. China's 14th Five-Year Plan includes explicit targets for phosphate reuse in the fertilizer sector.
On the innovation front, researchers are exploring biological phosphate-accumulating organisms — bacteria that store polyphosphate intracellularly under anaerobic-aerobic cycling conditions — as a low-energy route to enhanced biological phosphorus removal and recovery. Hydrothermal conversion of phosphate-laden sewage sludge into hydroxyapatite is another frontier attracting significant research funding. And digitally controlled smart crystallizers, capable of adapting in real time to fluctuating influent chemistry, are beginning to enter commercial pilots.
For industrial companies, the case for early adoption is compelling. Facilities that invest in phosphate recovery infrastructure now will be better positioned as regulatory constraints tighten, will benefit from first-mover advantages in the emerging market for recovered fertilizers, and will have a tangible sustainability story to tell investors and customers alike. The question is no longer whether to recover — it is how soon, and at what scale.
From Waste Stream to Value Stream
Industrial wastewater has long been framed as a liability — a costly byproduct requiring treatment and disposal. Phosphate recovery reframes that narrative entirely. In the dissolved phosphorus flowing through factory drains, there is a recoverable, saleable commodity; a contribution to food security; a reduction in eutrophication risk; and a step toward the circular economy that policymakers, investors, and consumers are demanding.
Upcycling industrial wastewater is not a distant aspiration. The technologies exist. The regulatory frameworks are taking shape. The economics are improving. What is needed now is the industrial will to treat every kilogram of discharged phosphate not as a waste management problem but as a resource management opportunity — one that sits, waiting, at the end of every production line.