Phosphoric Acid in Lithium Iron Phosphate (LFP) Battery Production: The Chemistry Powering the EV Revolution
The electric vehicle revolution is not just a story about motors and software; it is fundamentally a story about chemistry. At the heart of the fastest-growing battery technology in the world sits a molecule that has been a cornerstone of industrial chemistry for over a century: phosphoric acid (H₃PO₄). As lithium iron phosphate (LFP) batteries surge to dominate global EV production, the role of high-purity phosphoric acid has never been more strategically important.
What Is an LFP Battery, and Why Is It Winning?
Lithium iron phosphate batteries belong to the broader family of lithium-ion batteries, but they use lithium iron phosphate (LiFePO₄) as the cathode active material rather than the cobalt- or nickel-based chemistries that dominated the previous decade. This distinction matters enormously.
LFP batteries offer a compelling combination of properties: exceptional thermal stability, long cycle life (often exceeding 3,000–4,000 charge cycles), strong safety characteristics, and, critically, freedom from cobalt and nickel, two metals associated with volatile supply chains, high costs, and ethical sourcing concerns.
As a result, LFP has become the chemistry of choice for entry-level and mid-range EVs, electric buses, stationary energy storage systems, and increasingly, premium vehicles where range concerns are offset by competitive pricing. Tesla, BYD, CATL, and a growing roster of manufacturers have made LFP their default or primary chemistry. By 2023, LFP batteries accounted for well over 40% of global EV battery production, and that share continues to climb.
Behind every LFP cell, there is phosphoric acid.
The Chemistry: How Phosphoric Acid Becomes LiFePO₄
Understanding the role of phosphoric acid begins with understanding the synthesis of LiFePO₄ cathode material. The most widely used industrial process is the hydrothermal or co-precipitation synthesis, though solid-state and sol-gel routes are also employed. Regardless of the method, phosphoric acid (or its salts) serves as the essential phosphate source.
The Core Reaction
Controlling particle morphology, size distribution, and crystal structure is critical to the electrochemical performance of the final material. Iron must be maintained in its ferrous (Fe²⁺) state throughout the process to ensure the correct olivine crystal structure of LiFePO₄, as ferric iron (Fe³⁺) leads to impurities that degrade battery performance.
Phosphoric acid plays several roles simultaneously: it provides the phosphate ions (PO₄³⁻) that form the iron-phosphate backbone of the crystal lattice, contributes to pH regulation during synthesis, and in some routes functions as a mild reducing agent to help maintain the Fe²⁺ state.
The Olivine Structure: Why Phosphate Matters
The remarkable stability of LFP batteries is directly attributable to the olivine crystal structure of LiFePO₄, and that structure exists because of the strong covalent bonding within the phosphate (PO₄³⁻) polyanion. The P–O bond is exceptionally stable and does not readily release oxygen even under thermal abuse conditions — a stark contrast to layered oxide cathodes like NMC or NCA, which can release oxygen when overheated and contribute to thermal runaway.
In short, the phosphate group is not merely a structural component; it is the very reason LFP batteries are among the safest lithium-ion chemistries available. The quality and purity of the phosphoric acid used in synthesis directly determine the integrity of that phosphate backbone.
Purity: The Critical Variable
Not all phosphoric acid is equal. Industrial-grade phosphoric acid — widely used in fertilizers, food processing, and metal treatment — contains heavy metal impurities such as arsenic (As), cadmium (Cd), lead (Pb), fluoride (F⁻), and sulfate (SO₄²⁻) at levels that are acceptable for agricultural or construction applications but catastrophically problematic for battery production.
In LFP cathode synthesis, trace impurities can disrupt the olivine crystal lattice (reducing specific capacity and cycle stability), poison the iron reduction process, leading to Fe³⁺ contamination and capacity fade, introduce ionic impurities into the electrolyte, accelerating degradation, and create parasitic side reactions during charging and discharging.
Battery-grade phosphoric acid must typically meet specifications including arsenic content below 1 ppm, heavy metals (Pb, Cd, Hg) in the low ppb range, chloride and sulfate levels under 5–10 ppm, and iron content controlled within very tight limits. The phosphoric acid must also have a precisely known concentration — typically supplied as 85% w/w food or battery grade — to enable accurate stoichiometric control during synthesis.
Suppliers capable of providing phosphoric acid at these purity levels, with consistent batch-to-batch quality and reliable supply chain documentation, occupy a critical and increasingly strategic position in the battery materials supply chain.
The Supply Chain Dimension
The exponential growth of LFP battery production has created substantial and rapidly growing demand for battery-grade phosphoric acid. This demand exists within a broader supply landscape where phosphoric acid production has historically been dominated by the fertilizer industry, which requires far lower purity standards.
The production of battery-grade phosphoric acid typically involves an additional purification stage — often solvent extraction or ion exchange — applied to wet-process phosphoric acid derived from phosphate rock. This upgrading process is technically demanding, capital-intensive, and requires consistent quality management.
Key phosphoric acid production capacity is geographically concentrated, with major operations in China, Morocco (via phosphate rock from OCP Group), the United States, and parts of the Middle East. China currently dominates both phosphate rock processing and battery-grade phosphoric acid production, a position that has prompted significant concern and investment in supply chain diversification among European and North American battery manufacturers.
For battery manufacturers, procurement strategy around phosphoric acid increasingly resembles the strategic sourcing approaches more commonly associated with lithium or cobalt: long-term contracts, supplier qualification processes, and active engagement in upstream supply security.
Industrial-Scale Considerations: From Synthesis to Cell
Beyond cathode powder synthesis, phosphoric acid and phosphate compounds appear at several other points in the LFP battery manufacturing process, though the cathode synthesis stage represents by far the largest volume consumption.
In some electrolyte formulations, phosphate-based additives are used to form a stable solid-electrolyte interphase (SEI) on electrode surfaces. Lithium hexafluorophosphate (LiPF₆), the most common lithium-ion electrolyte salt, also involves phosphorus chemistry, though it is synthesised via different phosphorus precursors.
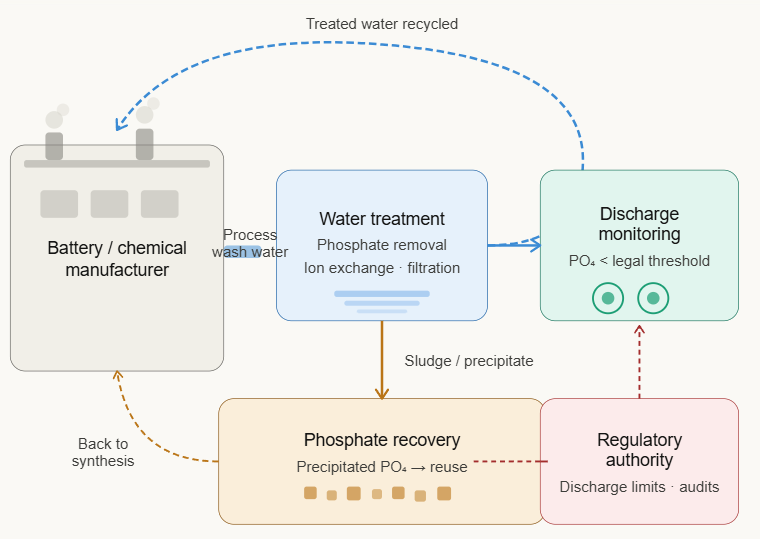
At the cell manufacturing scale, process water management is critical: residual phosphate in wash water from cathode synthesis must be carefully controlled to meet environmental discharge standards. Battery manufacturers and their chemical suppliers are under increasing regulatory pressure to demonstrate closed-loop water management and minimal phosphate discharge, particularly in regions with stringent water quality regulations.
LFP’s Role in the Energy Transition and What It Means for Phosphoric Acid Demand
The International Energy Agency and major battery market analysts project continued robust growth in LFP deployments through the end of the decade. Stationary energy storage — grid-scale batteries for renewable integration — is projected to grow even faster than automotive applications on a percentage basis, and LFP is the dominant chemistry for this segment due to its cycle life and cost characteristics.
Conservative estimates suggest global LFP cathode production could reach several million metric tons per year by 2030, with corresponding phosphoric acid demand measured in hundreds of thousands of metric tons of battery-grade material annually. This represents a structural shift in what has historically been a niche high-purity chemicals market.
For chemical suppliers with the technical capability to produce battery-grade phosphoric acid reliably and at scale, this growth trajectory represents a significant commercial opportunity. Qualification as a battery-grade phosphoric acid supplier to major LFP cathode manufacturers involves rigorous technical evaluation, quality audits, and often multi-year approval processes — creating meaningful barriers to entry and long-term supplier relationships for those who succeed.
The Road Ahead: Innovation in LFP Chemistry
Research into next-generation LFP and related materials is active and ongoing. Among the most significant developments is LMFP — lithium manganese iron phosphate (LiMnₓFe₋ₓPO₄) — which substitutes a proportion of iron with manganese to increase the operating voltage and thus the energy density of the cathode, addressing one of LFP’s traditional limitations. Phosphoric acid remains a central precursor in LMFP synthesis, with the added complexity of managing manganese chemistry alongside iron.
Additionally, advances in carbon-coating technology — in which LiFePO₄ particles are coated with a thin layer of conductive carbon to overcome the material’s intrinsically low electronic conductivity — continue to improve LFP performance. The quality of the initial phosphate source has been shown to influence carbon-coating uniformity, reinforcing the importance of consistently high-purity precursors.
Conclusion: A Foundational Chemistry for a Cleaner Future
Phosphoric acid may not feature in headlines about the EV revolution, but it is indispensable to it. Every LFP battery cell in every electric bus, electric car, and grid storage installation carries within it the phosphate chemistry that provides its safety, stability, and longevity — and that chemistry begins with phosphoric acid.
As the global battery industry scales at an unprecedented pace, the importance of a reliable, high-quality phosphoric acid supply will only grow. At DECACHEM, we understand the critical role that specialty and industrial chemicals play in enabling the technologies that define the energy transition. Supplying the right chemistry, at the right purity, at the right time, is not just a commercial proposition — it is a contribution to the infrastructure of a more sustainable world.
Interested in high-purity phosphoric acid for battery or advanced materials applications? Contact the DECACHEM team to discuss your requirements and explore how we can support your supply chain.